
Recombinant expression, purification and PEGylation of Paneth cell peptide (cryptdin-2) with value added attributes against Staphylococcus aureus | Scientific Reports
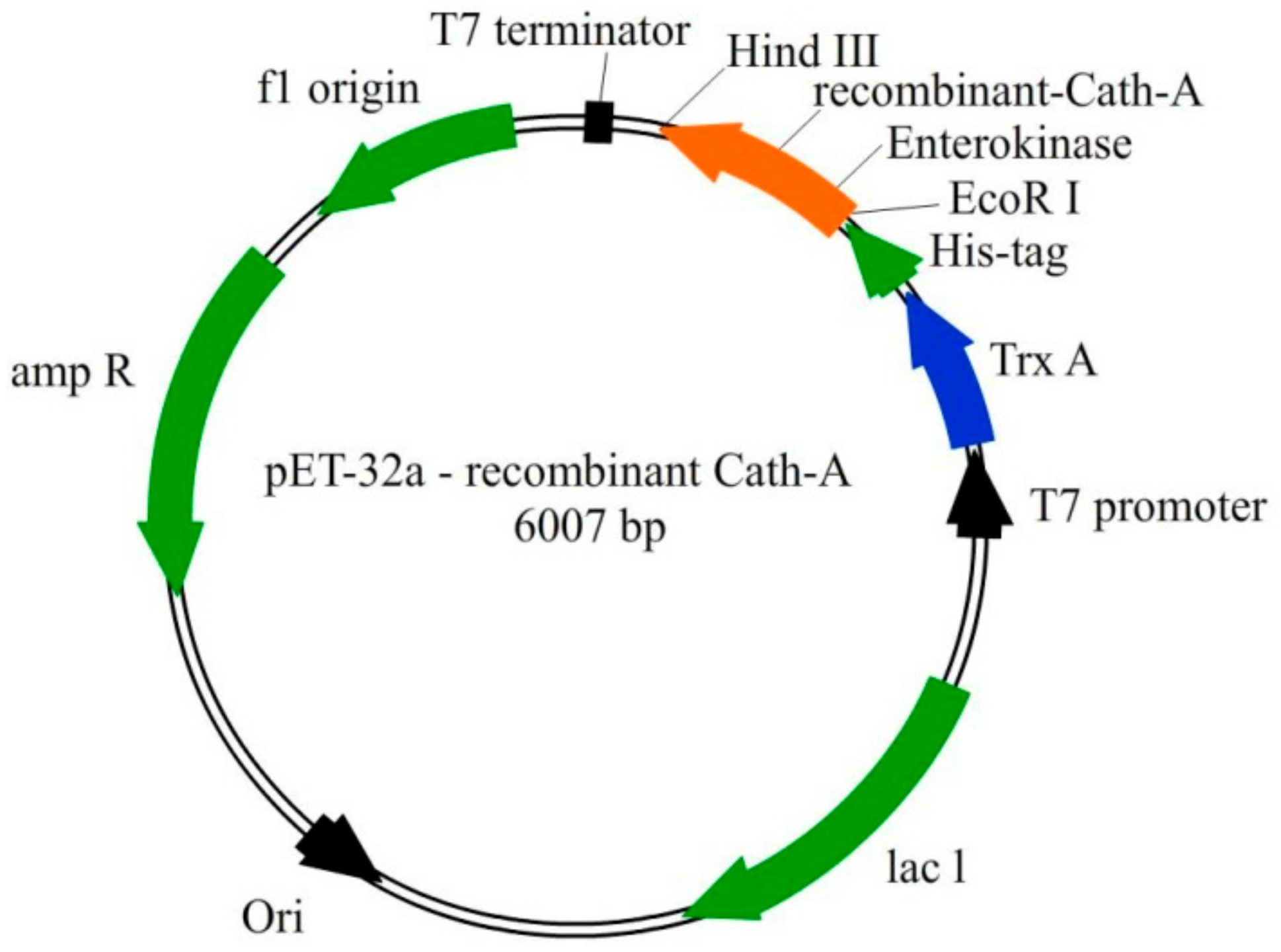
Biomolecules | Free Full-Text | A Recombinant Snake Cathelicidin Derivative Peptide: Antibiofilm Properties and Expression in Escherichia coli

Structural Basis for Target Protein Recognition by the Protein Disulfide Reductase Thioredoxin: Structure

Two-step metal affinity purification of double-tagged (NusA–His6) fusion proteins | Nature Protocols

Thioredoxin/TRX Protein Overview: Sequence, Structure, Function and Protein Interaction | Sino Biological
Protein Aggregation Formed by Recombinant cp19k Homologue of Balanus albicostatus Combined with an 18 kDa N-Terminus Encoded by pET-32a(+) Plasmid Having Adhesion Strength Comparable to Several Commercial Glues | PLOS ONE

SciELO - Brasil - Expression, Purification and Functional Assessment of Smallest Isoform of Human Interleukin-24 in <i>Escherichia coli</i> Expression, Purification and Functional Assessment of Smallest Isoform of Human Interleukin-24 in <i>Escherichia ...

Scheme of the recombinant proteins as produced upon cloning in pET32b... | Download Scientific Diagram
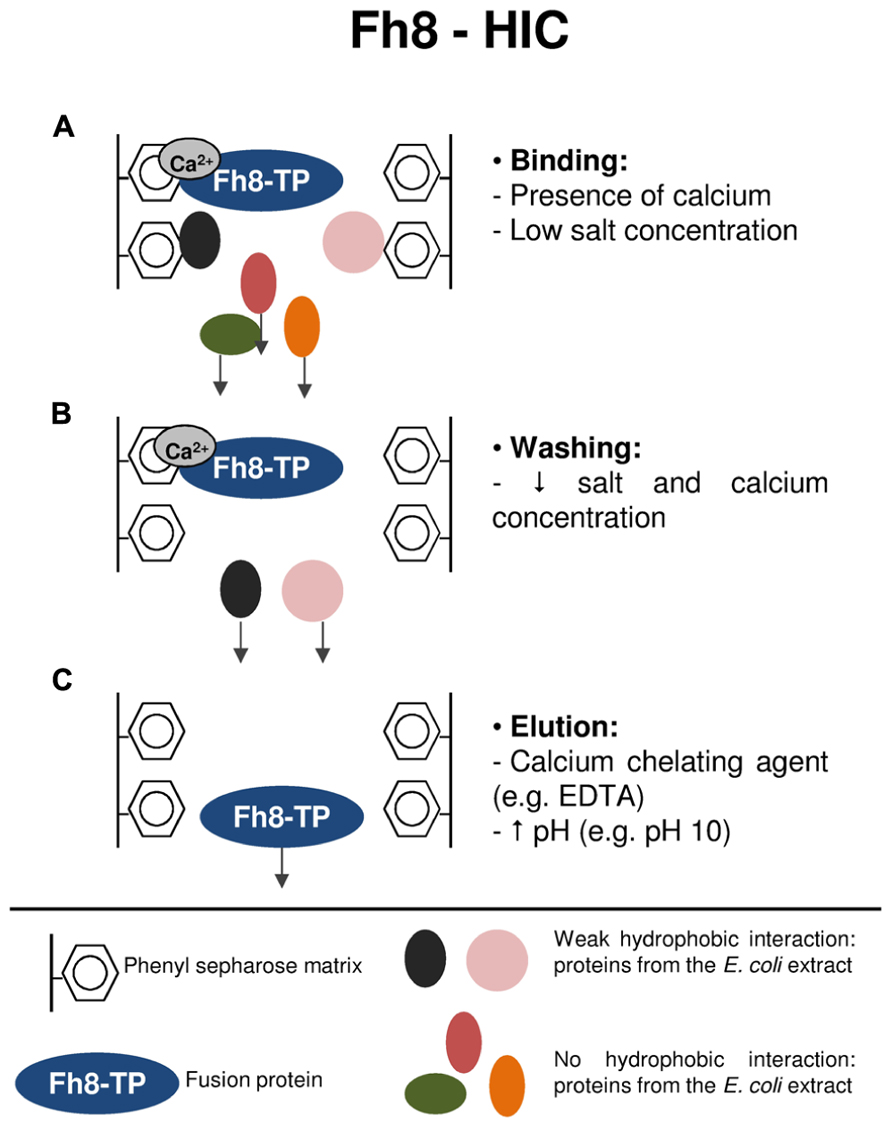
Frontiers | Fusion tags for protein solubility, purification and immunogenicity in Escherichia coli: the novel Fh8 system















![Recombinant Anti-Thioredoxin / TRX antibody [EPR6111] (ab109385) | Abcam Recombinant Anti-Thioredoxin / TRX antibody [EPR6111] (ab109385) | Abcam](https://www.abcam.com/ps/products/109/ab109385/Images/ab109385-468757-anti-thioredoxin-trx-antibody-epr6111-westernblot-thioredoxin-recombinant-protein-human.jpg)