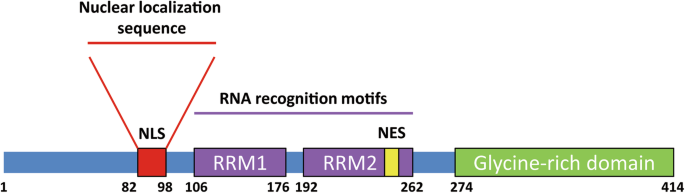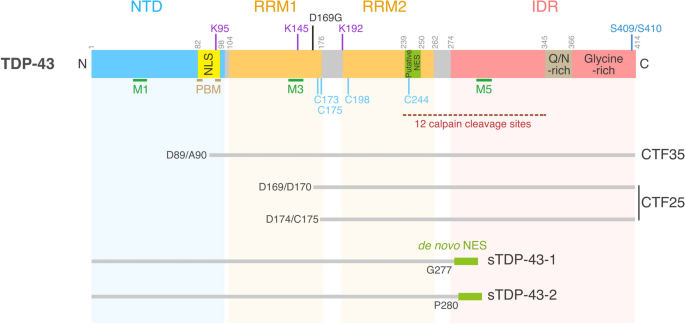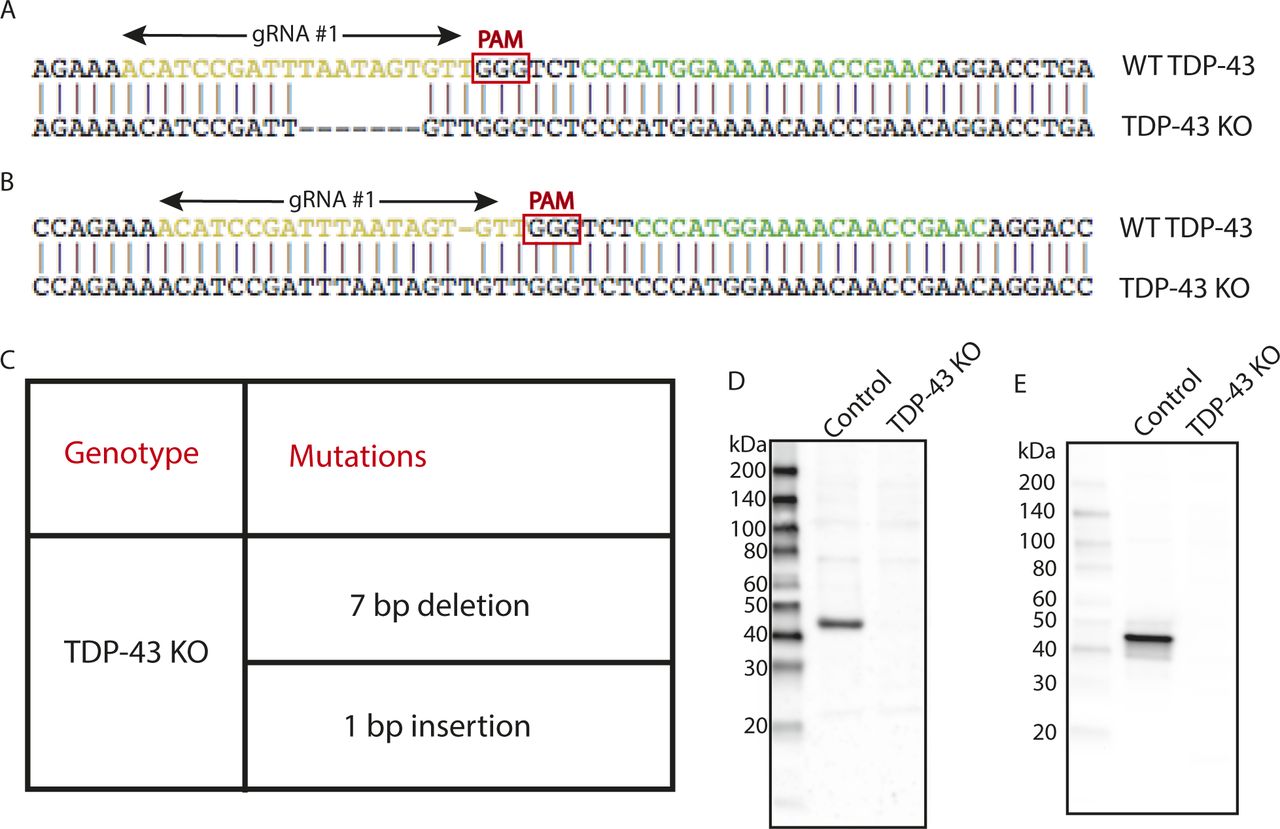
Pleiotropic requirements for human TDP-43 in the regulation of cell and organelle homeostasis | Life Science Alliance

TDP-43-mediated neurodegeneration: towards a loss-of-function hypothesis?: Trends in Molecular Medicine

A) Amino acid sequence of Transactive response DNA-binding protein 43... | Download Scientific Diagram
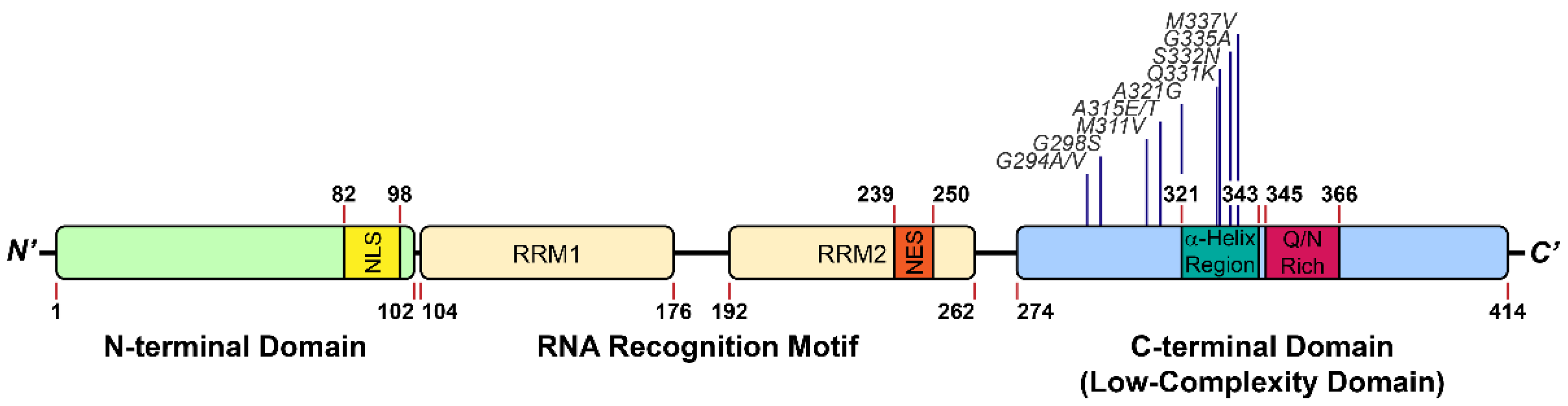
IJMS | Free Full-Text | The Different Faces of the TDP-43 Low-Complexity Domain: The Formation of Liquid Droplets and Amyloid Fibrils

TDP-43 N terminus encodes a novel ubiquitin-like fold and its unfolded form in equilibrium that can be shifted by binding to ssDNA | PNAS

Domain structures and assembly of TDP-43 proteins. (A) TDP-43 has two... | Download Scientific Diagram
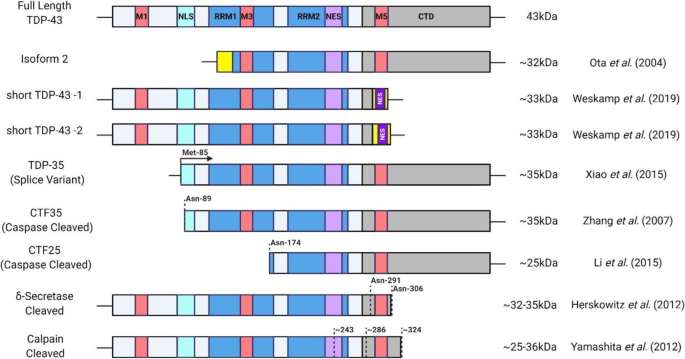
The role of TDP-43 mislocalization in amyotrophic lateral sclerosis | Molecular Neurodegeneration | Full Text

Mice with endogenous TDP‐43 mutations exhibit gain of splicing function and characteristics of amyotrophic lateral sclerosis | The EMBO Journal
A. Schematic representation of the primary structural architecture of... | Download Scientific Diagram

Pathogenic Mutation of TDP-43 Impairs RNA Processing in a Cell Type-Specific Manner: Implications for the Pathogenesis of ALS/FTLD | eNeuro

Structural analysis of disease-related TDP-43 D169G mutation: linking enhanced stability and caspase cleavage efficiency to protein accumulation | Scientific Reports
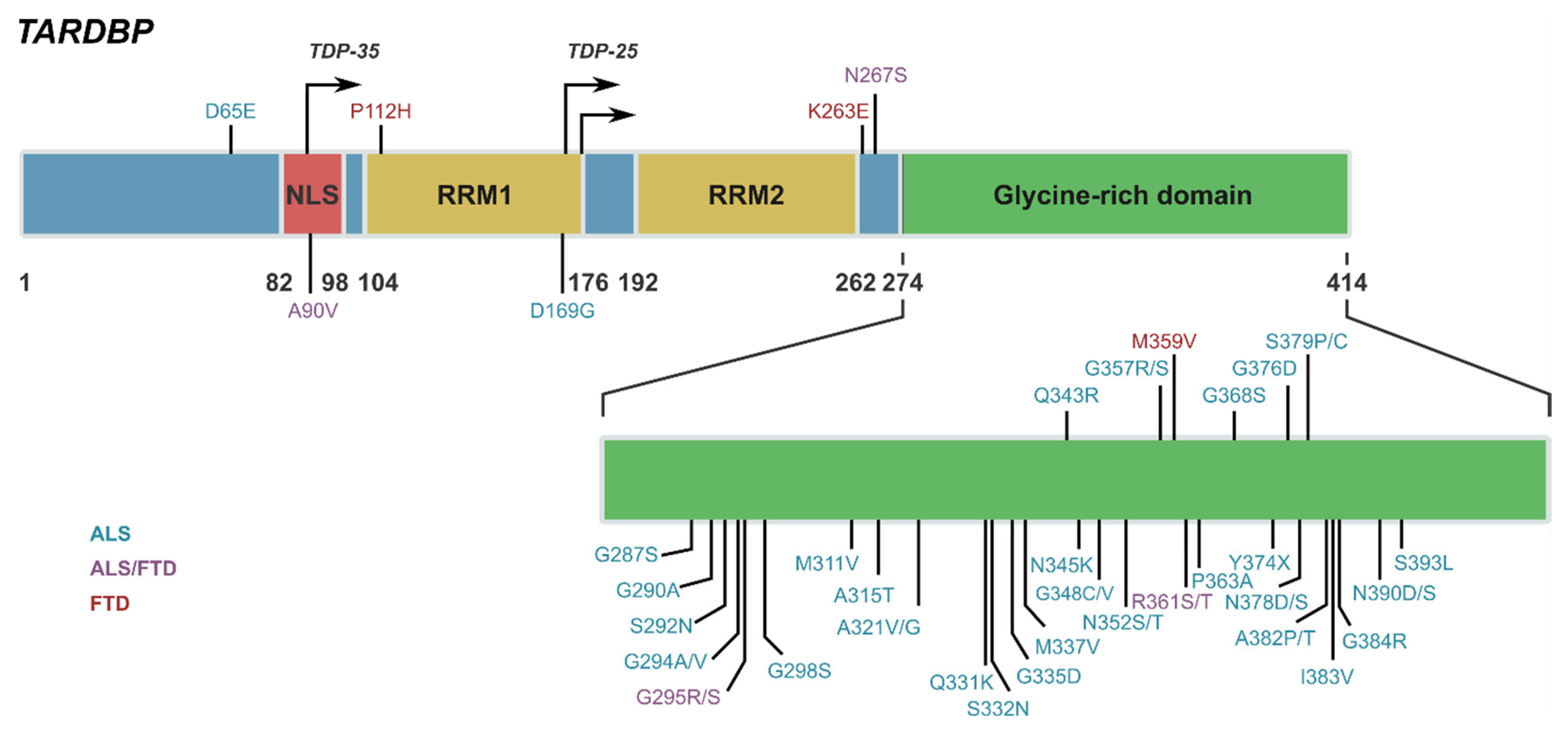
IJMS | Free Full-Text | TDP-43 and Inflammation: Implications for Amyotrophic Lateral Sclerosis and Frontotemporal Dementia

ALS-linked TDP-43 mutations interfere with the recruitment of RNA recognition motifs to G-quadruplex RNA | Scientific Reports

TDP-43 functions and pathogenic mechanisms implicated in TDP-43 proteinopathies: Trends in Molecular Medicine

Mice with endogenous TDP‐43 mutations exhibit gain of splicing function and characteristics of amyotrophic lateral sclerosis | The EMBO Journal

Strategies in the design and development of (TAR) DNA-binding protein 43 ( TDP-43) binding ligands - ScienceDirect