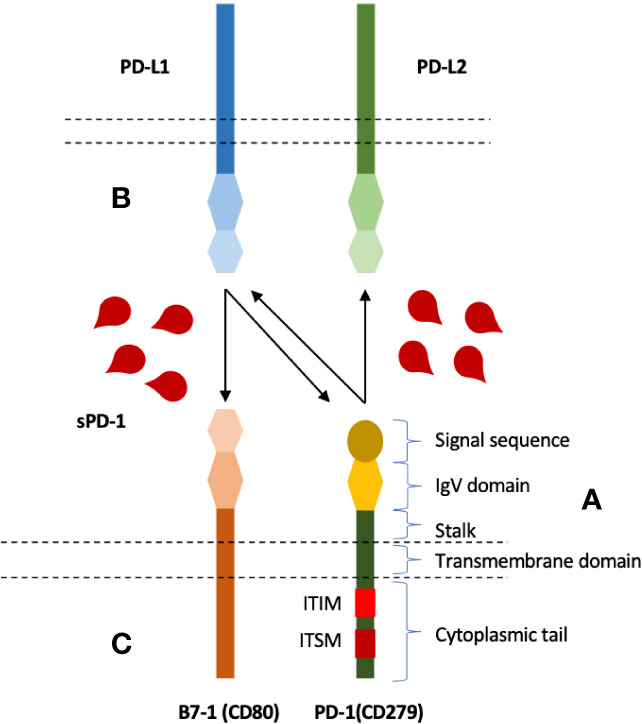
Crystal Structure of Pembrolizumab. The figure shows the amino acid... | Download Scientific Diagram

Whole-genome sequencing identifies responders to Pembrolizumab in relapse/refractory natural-killer/T cell lymphoma | Leukemia

The right Timing, right combination, right sequence, and right delivery for Cancer immunotherapy - ScienceDirect
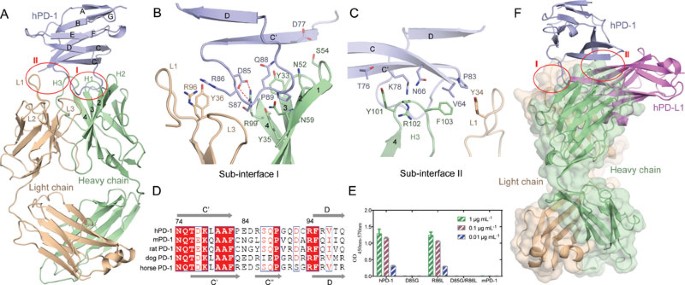
Structural basis for blocking PD-1-mediated immune suppression by therapeutic antibody pembrolizumab | Cell Research

Immunotherapy and radiation therapy sequencing: State of the data on timing, efficacy, and safety - Williamson - 2021 - Cancer - Wiley Online Library

High-resolution crystal structure of the therapeutic antibody pembrolizumab bound to the human PD-1 | Scientific Reports

WO2018204343A1 - Stable formulations of anti-ctla4 antibodies alone and in combination with programmed death receptor 1 (pd-1) antibodies and methods of use thereof - Google Patents

Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361): a randomised, open-label, phase 3 trial - The Lancet Oncology

Biomedicines | Free Full-Text | Analysis of Pembrolizumab in Human Plasma by LC-MS/HRMS. Method Validation and Comparison with Elisa
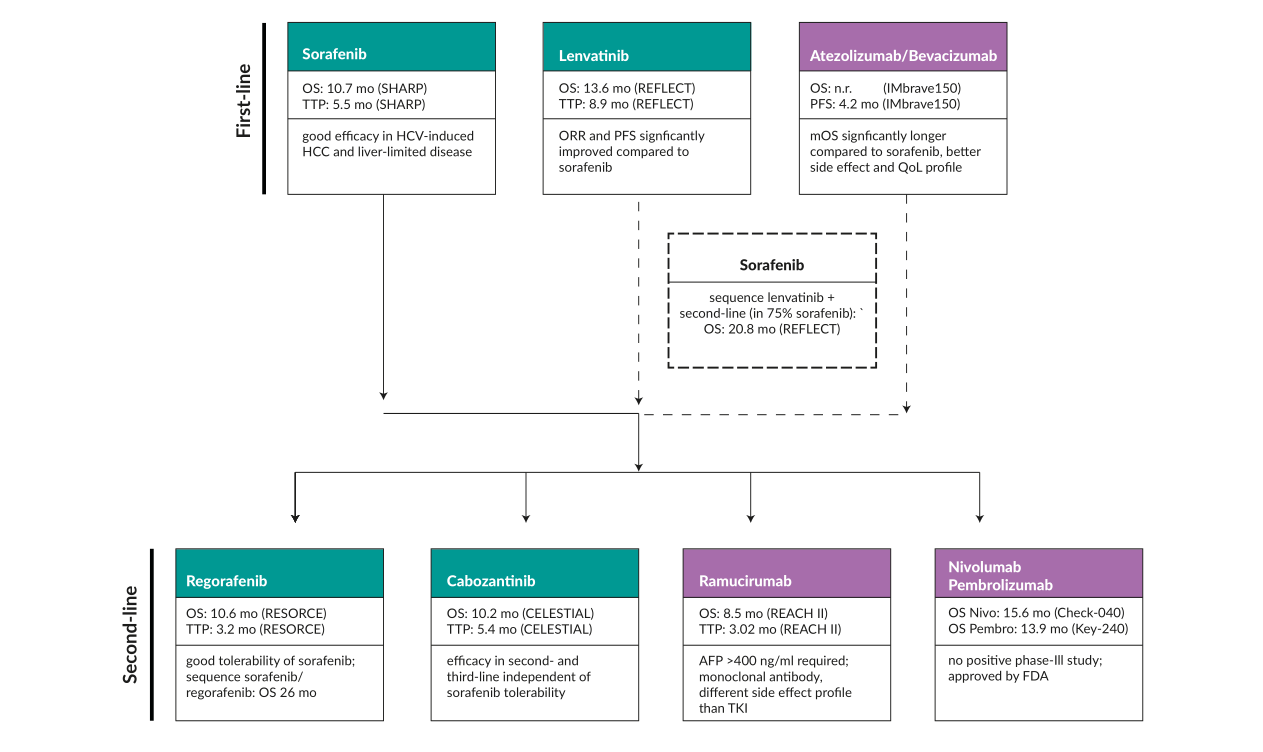
The Evolving Systemic Treatment for Advanced Hepatocellular Carcinoma | Published in healthbook TIMES Oncology Hematology
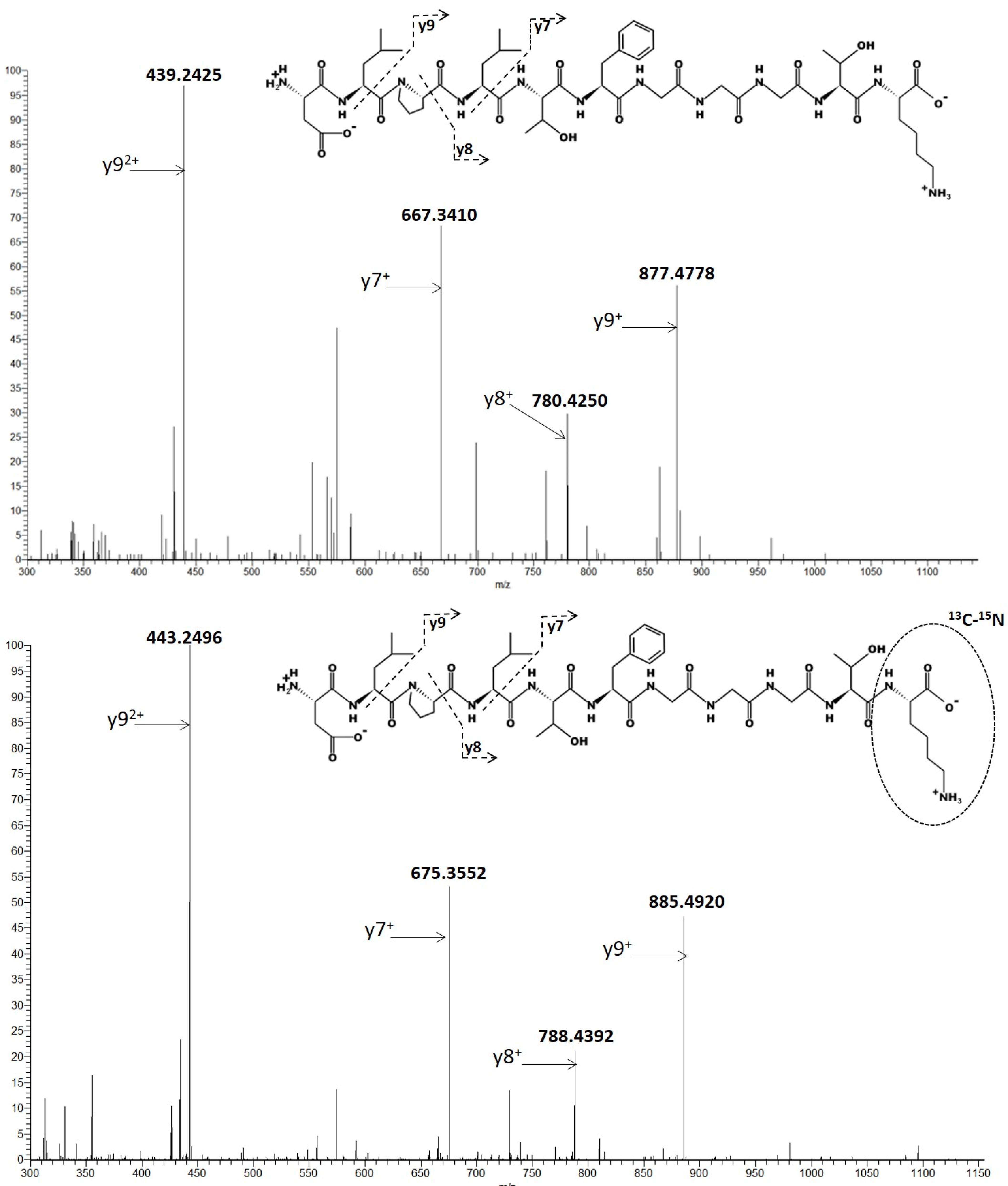
Biomedicines | Free Full-Text | Analysis of Pembrolizumab in Human Plasma by LC-MS/HRMS. Method Validation and Comparison with Elisa

Sequential treatment of progressive multifocal leukoencephalopathy with intravenous immunoglobulins and pembrolizumab | springermedizin.de


.png)