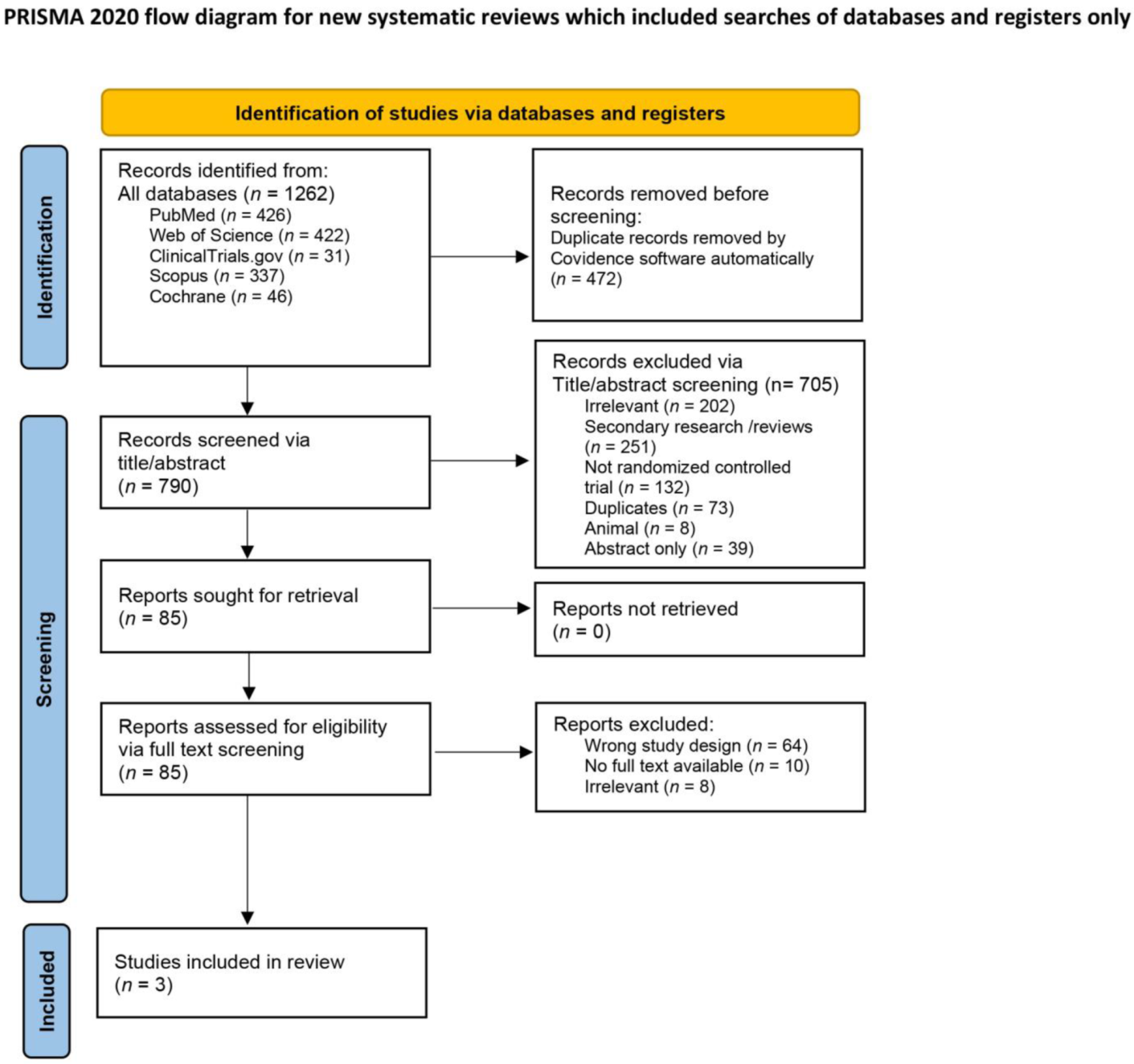
Medicina | Free Full-Text | The Safety and Efficacy of Nusinersen in the Treatment of Spinal Muscular Atrophy: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
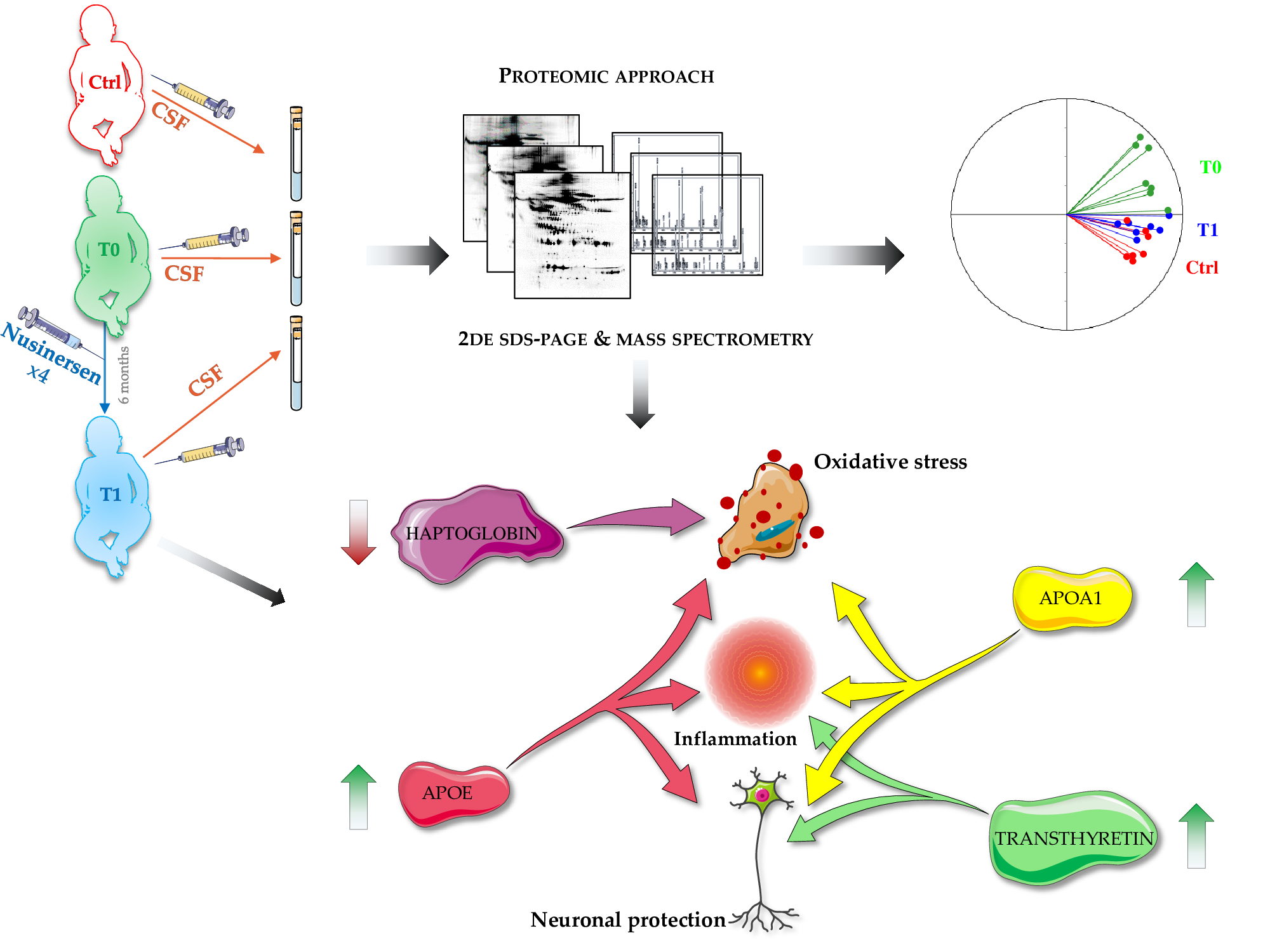
IJMS | Free Full-Text | Nusinersen Modulates Proteomics Profiles of Cerebrospinal Fluid in Spinal Muscular Atrophy Type 1 Patients
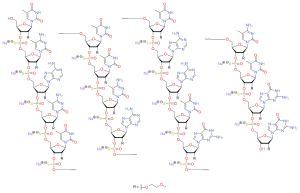
Antisense oligonucleotides (ASOs) in clinical use. (A) Nusinersen binds... | Download Scientific Diagram
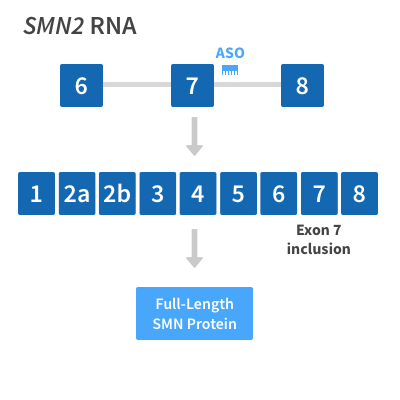
Nusinersen (Spinraza®) – Spinal Muscular Atrophy (SMA) | National Institute of Neurological Disorders and Stroke
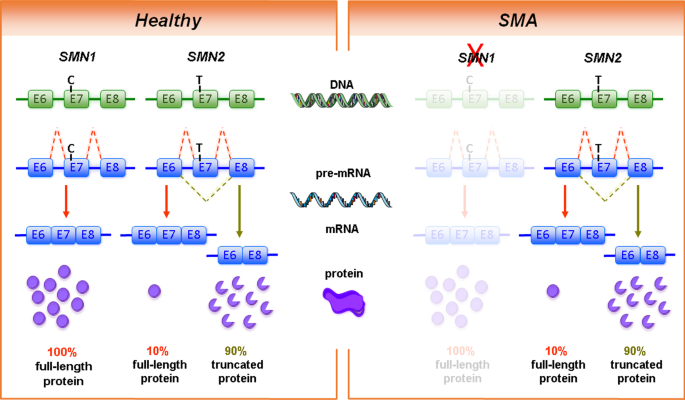
Therapy development for spinal muscular atrophy: perspectives for muscular dystrophies and neurodegenerative disorders | Neurological Research and Practice | Full Text

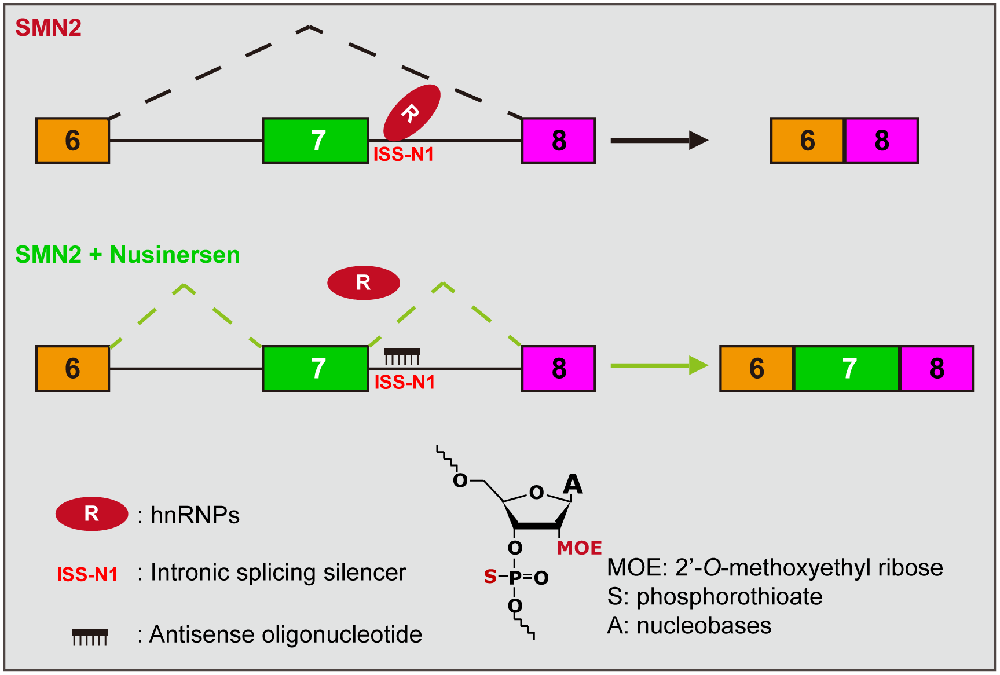
How the discovery of ISS-N1 led to the first medical therapy for spinal muscular atrophy | Gene Therapy

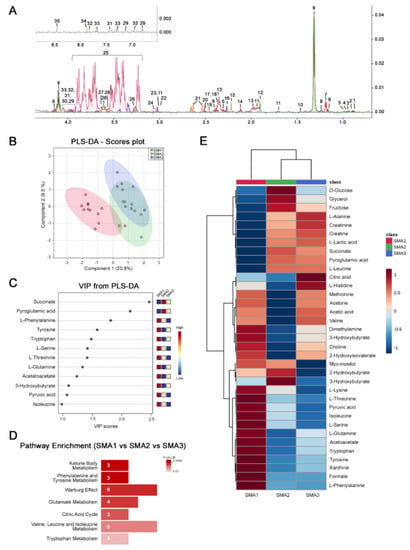
Biomolecules | Free Full-Text | Nusinersen Induces Disease-Severity-Specific Neurometabolic Effects in Spinal Muscular Atrophy
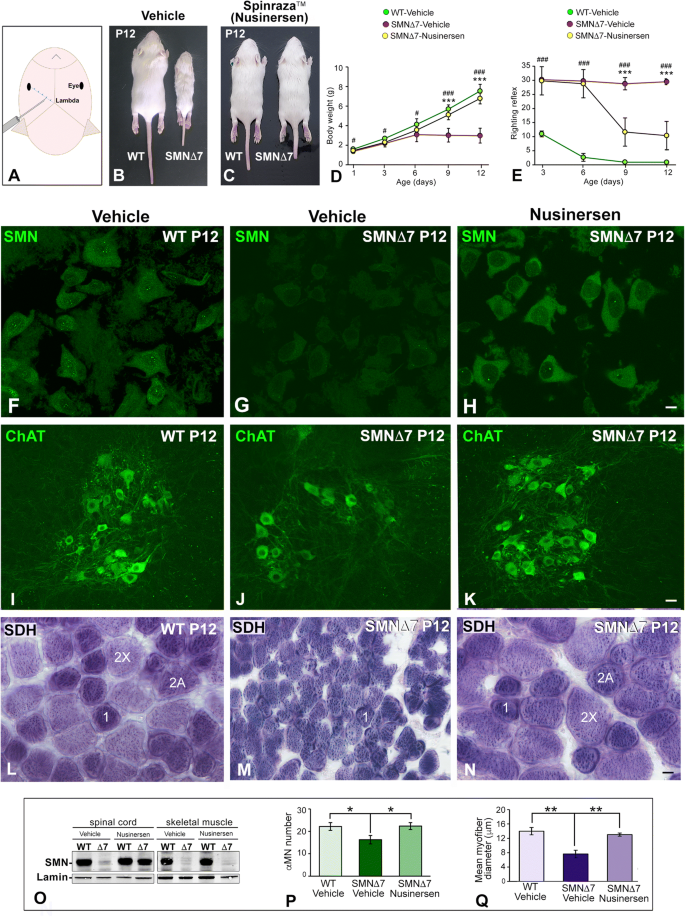
Nusinersen ameliorates motor function and prevents motoneuron Cajal body disassembly and abnormal poly(A) RNA distribution in a SMA mouse model | Scientific Reports

Treatment of infantile-onset spinal muscular atrophy with nusinersen: a phase 2, open-label, dose-escalation study - The Lancet
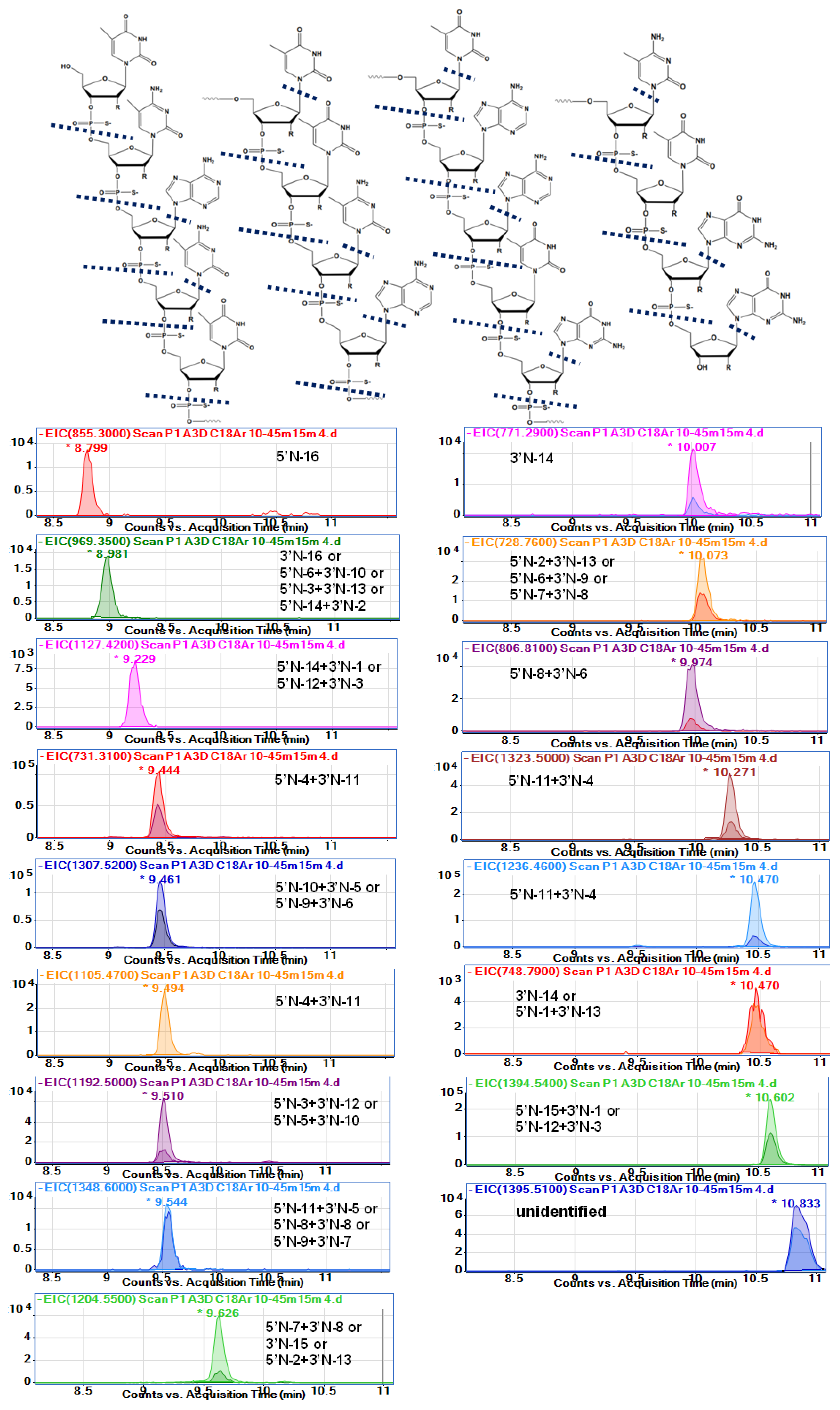
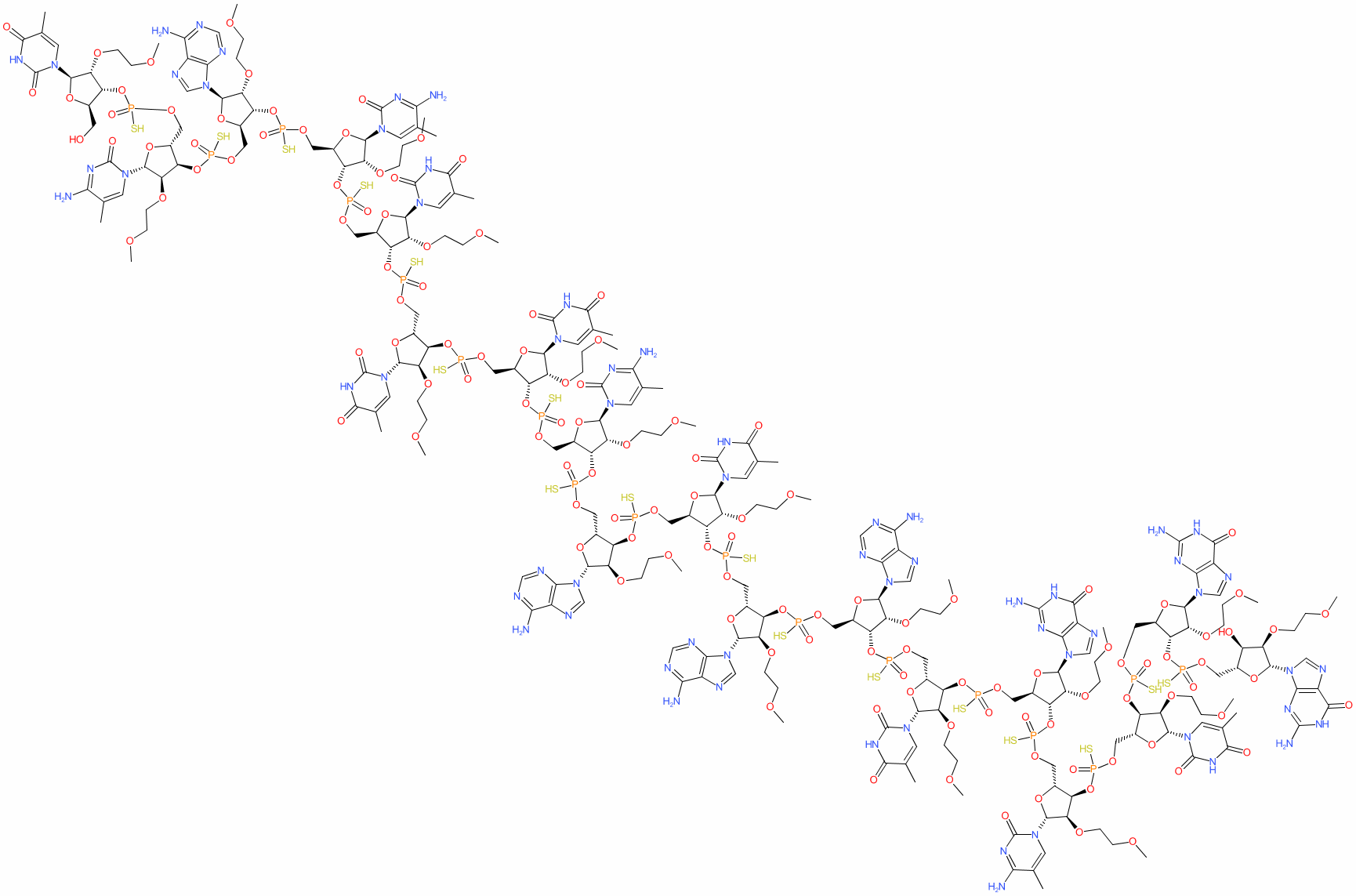
IJMS | Free Full-Text | Development of the Method for Nusinersen and Its Metabolites Identification in the Serum Samples of Children Treated with Spinraza for Spinal Muscular Atrophy