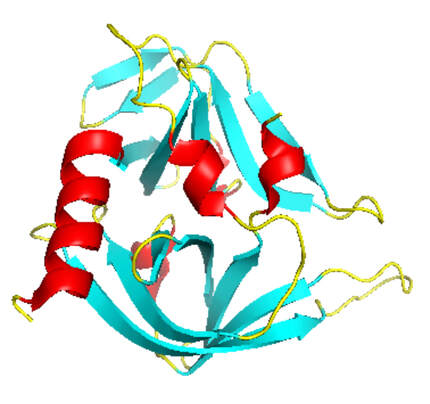
A) Three tags, i.e. 10x-His, FLAG and HA, and one proteolytic site for... | Download Scientific Diagram

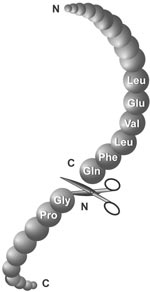
Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology

Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology

Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE

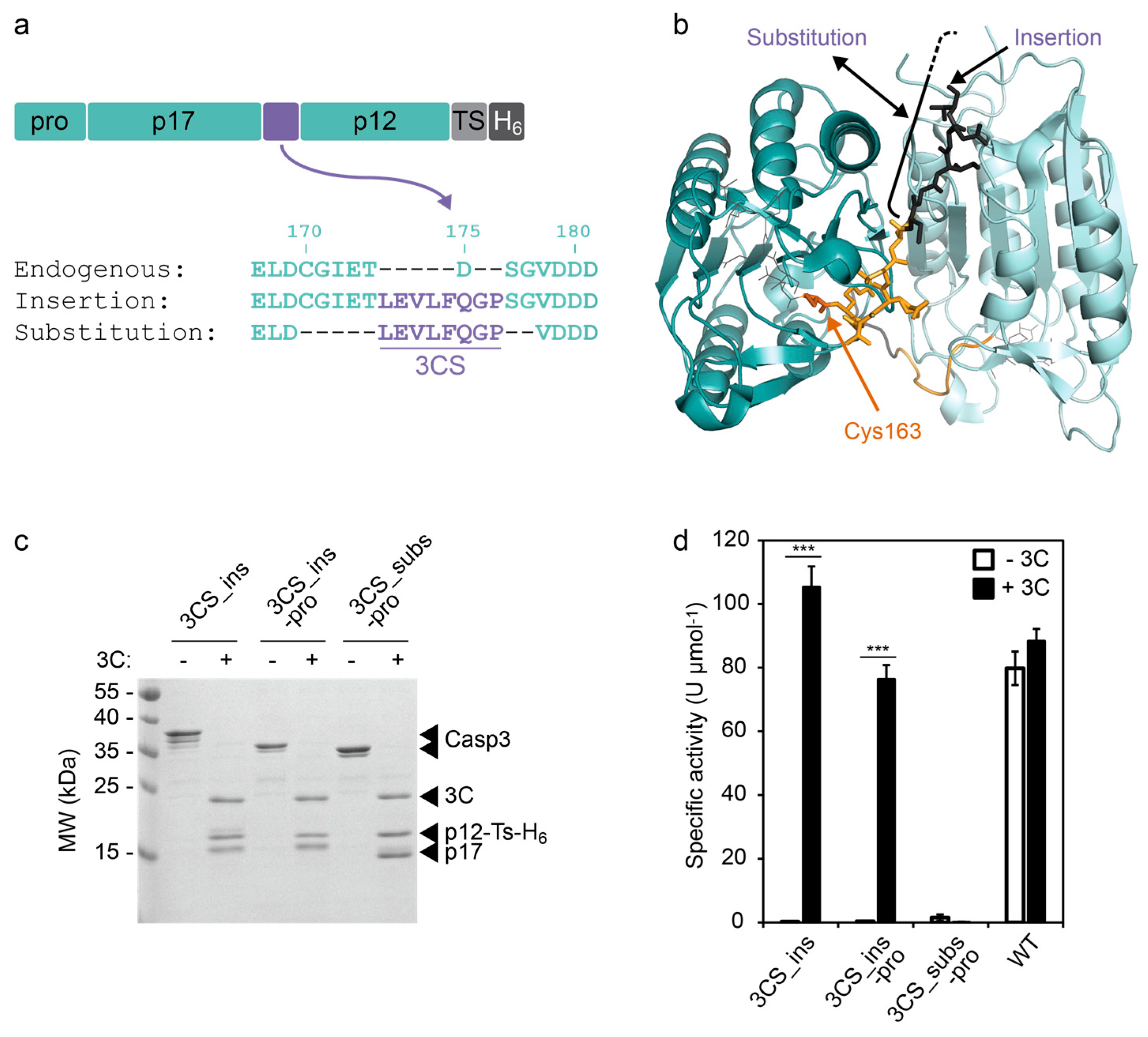
A split protease-E. coli ClpXP system quantifies protein–protein interactions in Escherichia coli cells | Communications Biology
A Versatile Strategy for Production of Membrane Proteins with Diverse Topologies: Application to Investigation of Bacterial Homologues of Human Divalent Metal Ion and Nucleoside Transporters | PLOS ONE
SDS-PAGE analysis of cleavage of His 8 -MBP-HRV 3C-domain by the HRV 3C... | Download Scientific Diagram
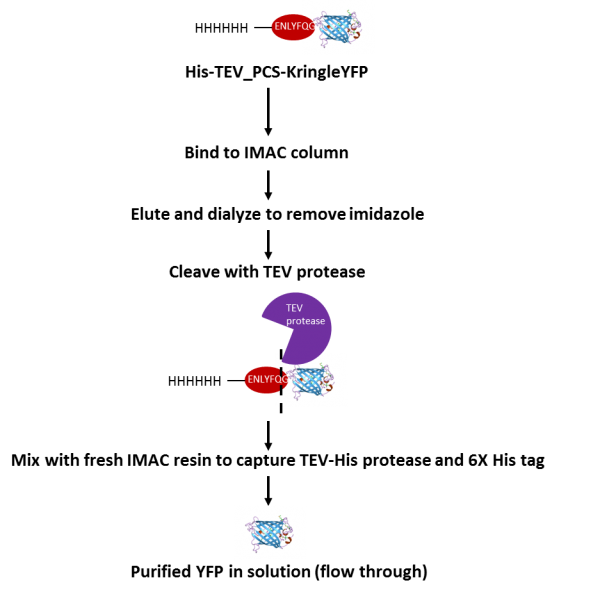
Enzymes - Tobacco Etch Virus (TEV) and Human RhinoVirus (HRV3C) Cysteine Proteases in Vectors | ATUM - ATUM