A simple solid phase, peptide-based fluorescent assay for the efficient and universal screening of HRV 3C protease inhibitors - ScienceDirect
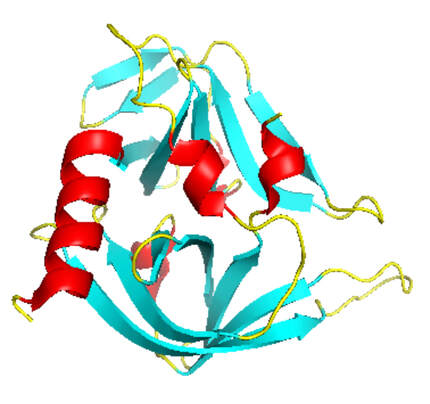
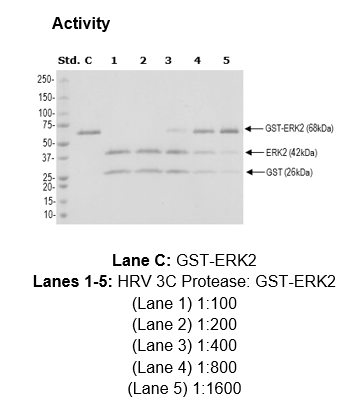
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE

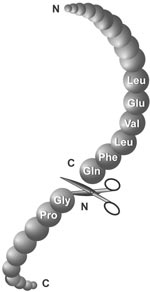
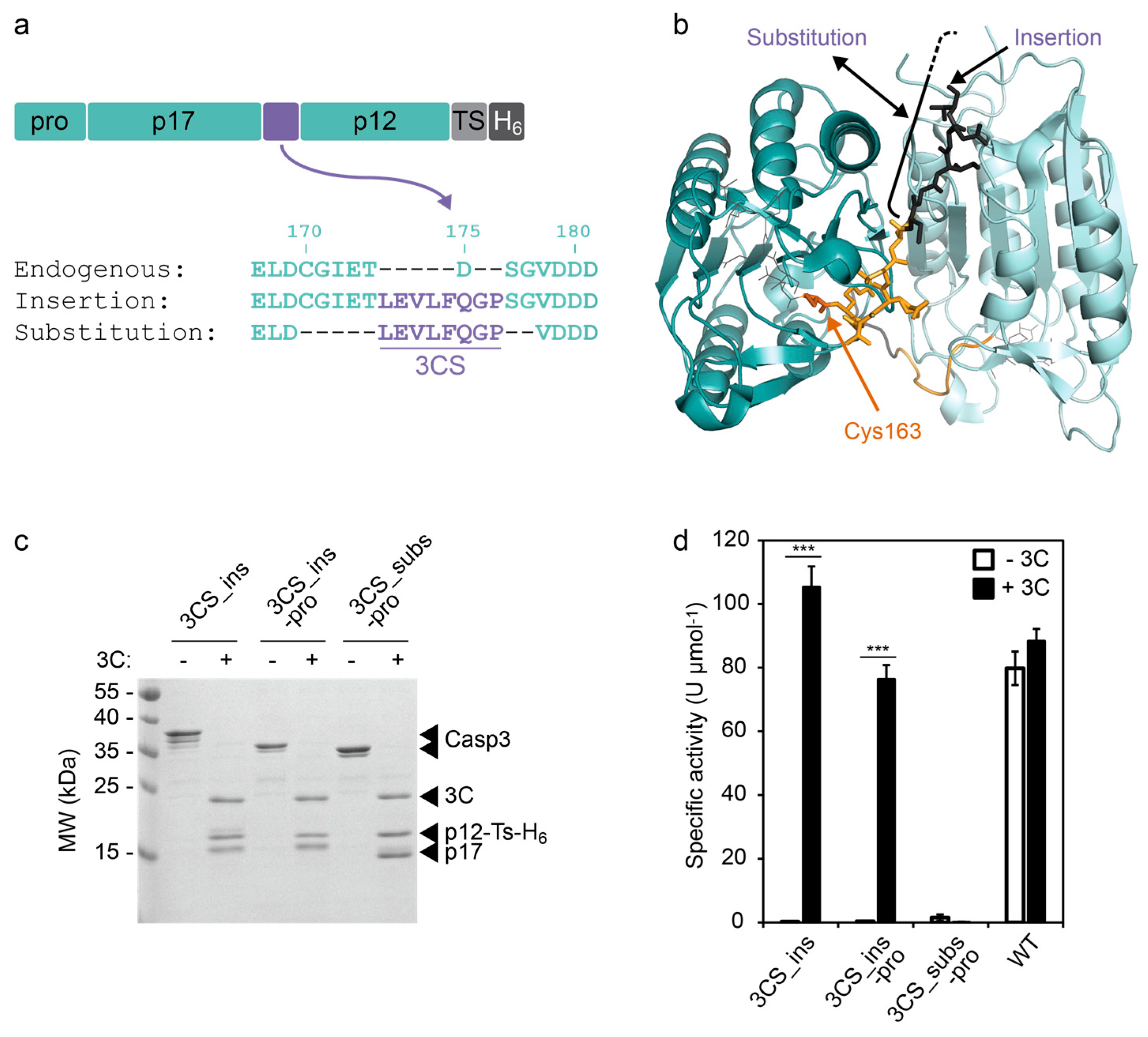
NT*-HRV3CP: An optimized construct of human rhinovirus 14 3C protease for high-yield expression and fast affinity-tag cleavage - ScienceDirect
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE
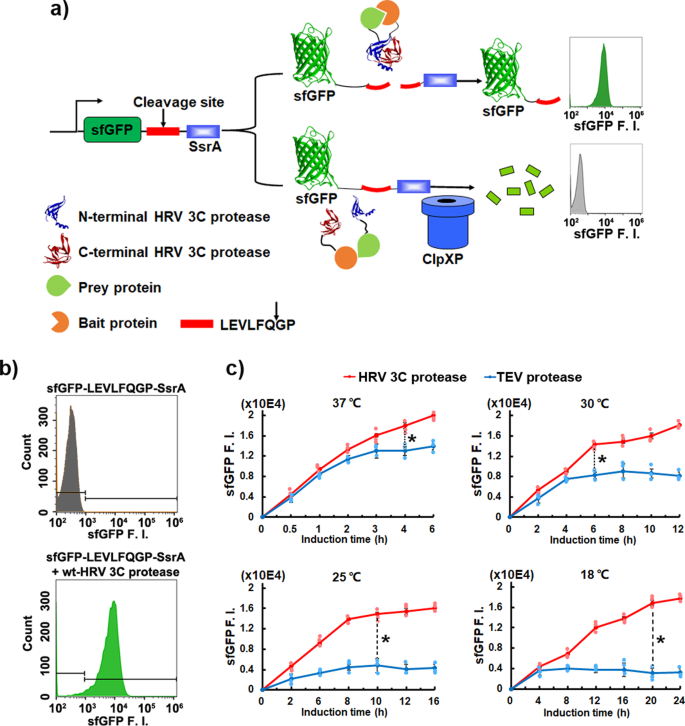
A split protease-E. coli ClpXP system quantifies protein–protein interactions in Escherichia coli cells | Communications Biology

Human Rhinovirus 3C protease cleaves RIPK1, concurrent with caspase 8 activation | Scientific Reports