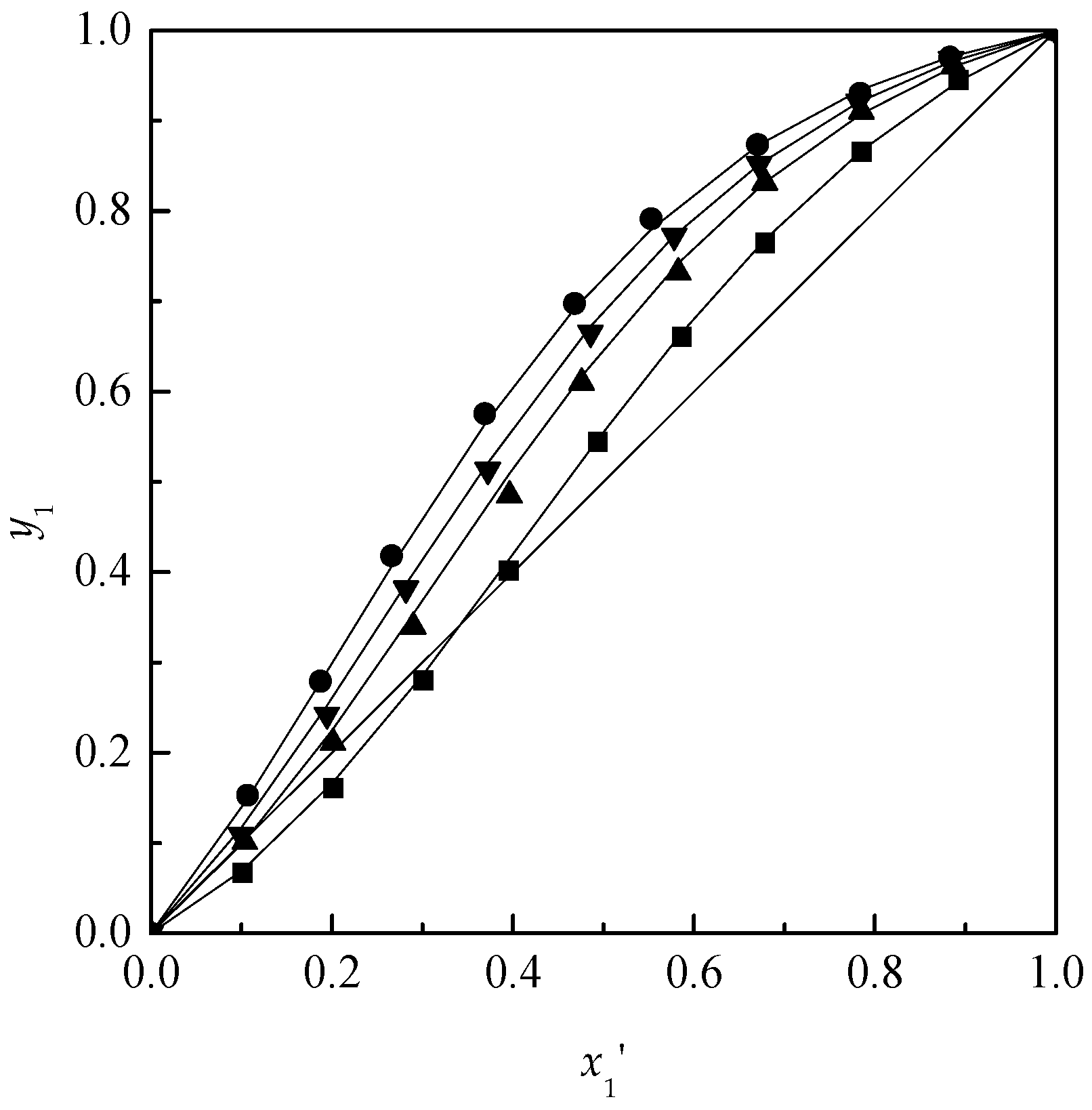
Comparison of extractive distillation and pressure-swing distillation for acetone/chloroform separation - ScienceDirect

Enhanced Mobility of Poly(3-hexylthiophene) Transistors by Spin-Coating from High-Boiling-Point Solvents | Chemistry of Materials

Compact organic liquid dielectric resonator antenna for air pressure sensing using soft material | Scientific Reports

Potential Use of Limonene as an Alternative Solvent for Extraction of Gutta-Percha from Eucommia ulmoides | ACS Sustainable Chemistry & Engineering

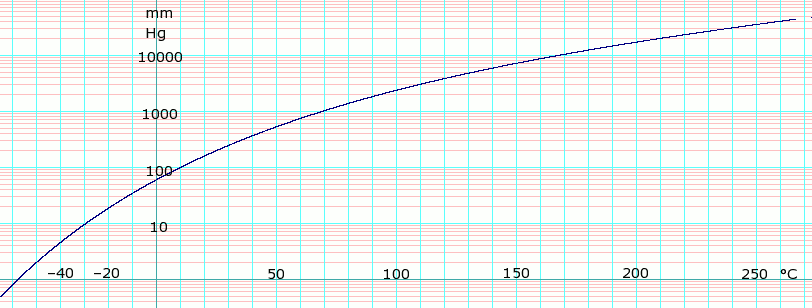
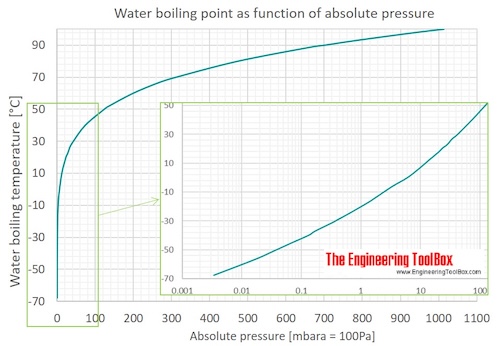
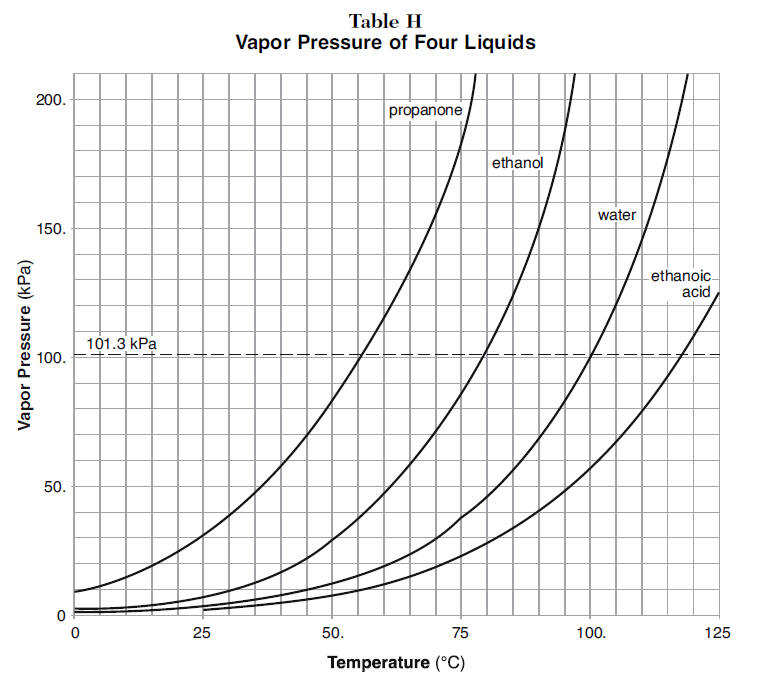
Use the figure below to determine the boiling point of -Chloroform at 80 kPa -Ethanol at 20kPa -Ethanol at - Brainly.com

Comparison of extractive distillation and pressure-swing distillation for acetone/chloroform separation - ScienceDirect
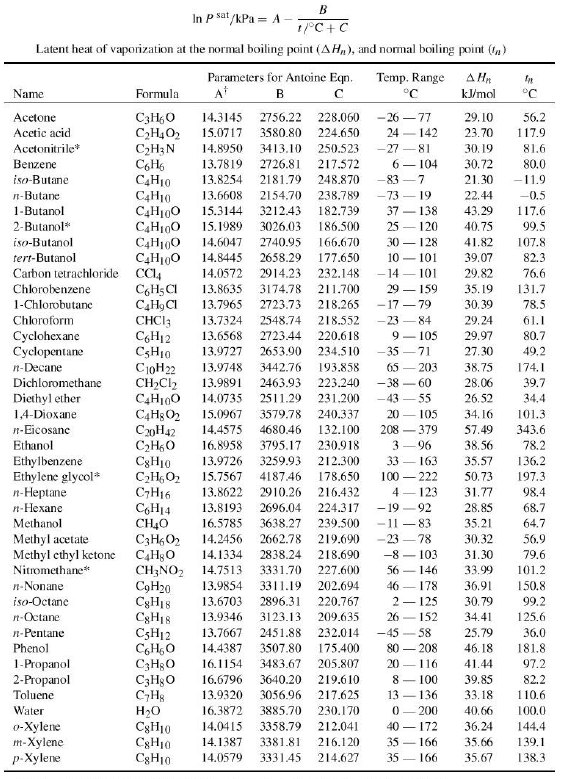
![Density, Viscosity, Vapor−Liquid Equilibrium, and Excess Molar Enthalpy of [ Chloroform + Methyl tert-Butyl Ether] | Journal of Chemical & Engineering Data Density, Viscosity, Vapor−Liquid Equilibrium, and Excess Molar Enthalpy of [ Chloroform + Methyl tert-Butyl Ether] | Journal of Chemical & Engineering Data](https://pubs.acs.org/cms/10.1021/je100821g/asset/images/medium/je-2010-00821g_0003.gif)
Density, Viscosity, Vapor−Liquid Equilibrium, and Excess Molar Enthalpy of [ Chloroform + Methyl tert-Butyl Ether] | Journal of Chemical & Engineering Data