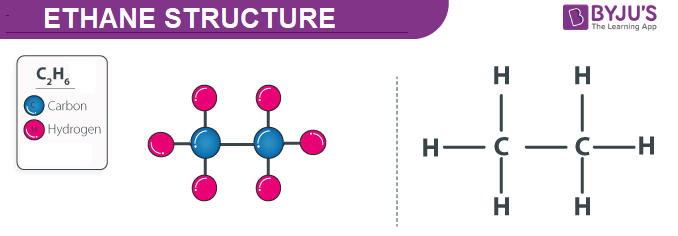
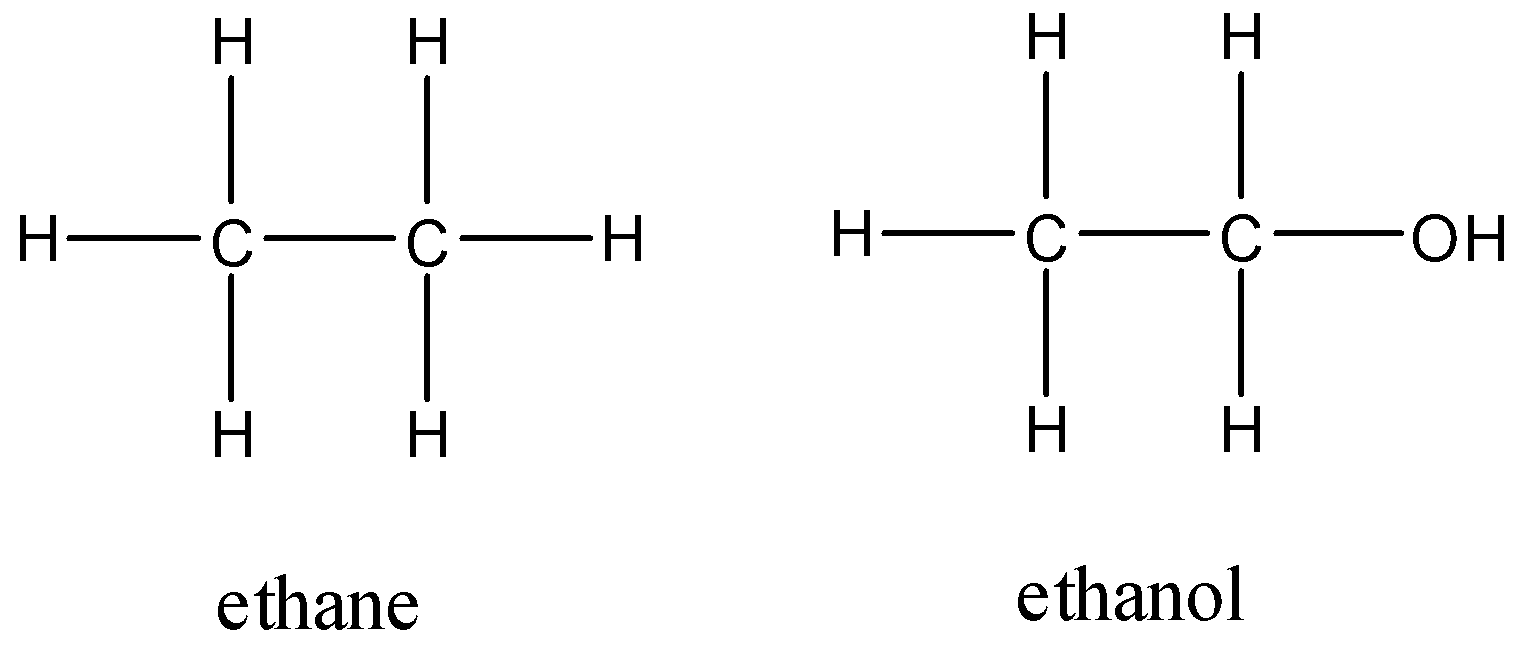
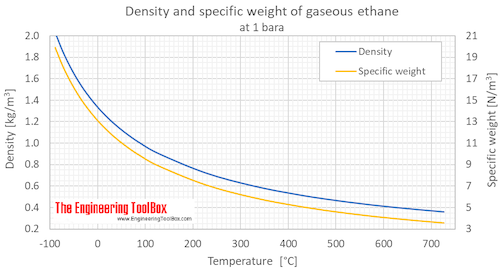
SOLVED: Claim: Propane (CH;CHzCHa) has a higher boiling point than ethane (CH;CHz): Evidence: Propane contains more bonds Propane contains larger electron cloud. Reasoning: More bonds require more energy to break so the

SOLVED:Pick the compound with the highest boiling point in each pair. Explain your reasoning. a. CH3OH or CH3SH b. CH3OCH3 or CH3CH2OH c. CH4 or CH3CH3

SOLVED: Arrange these compounds in order of increasing boiling point. Explain your reasoning. a. CH b. CH3CH3 c. CH3CH2Cl d. CH3CH2OH

The increasing order of the boiling points for the following compounds is : (I)C2H5OH (II)C2H5Cl (III)C2H5CH3 (IV)C2H5OCH3


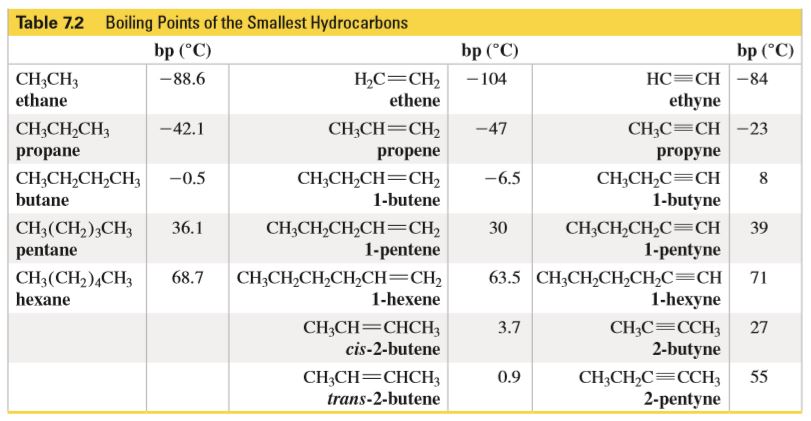
.PNG)