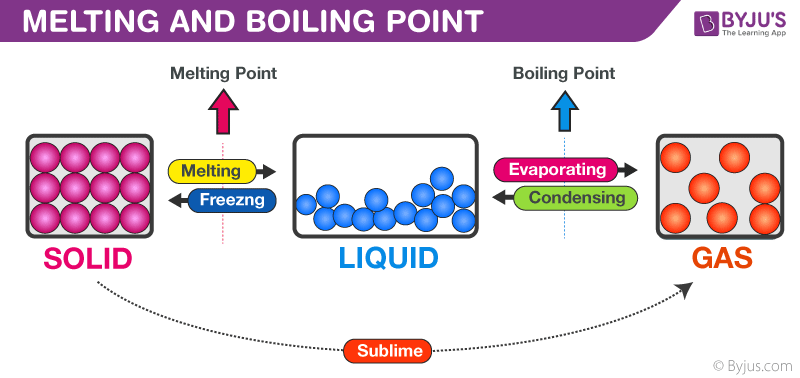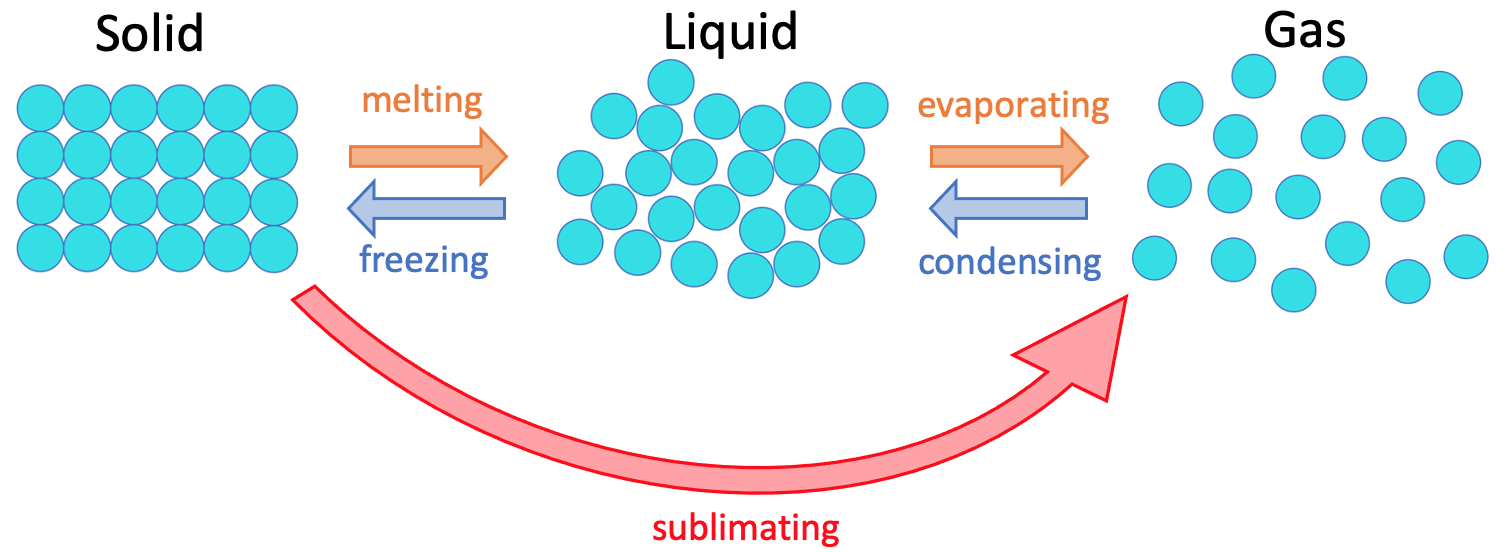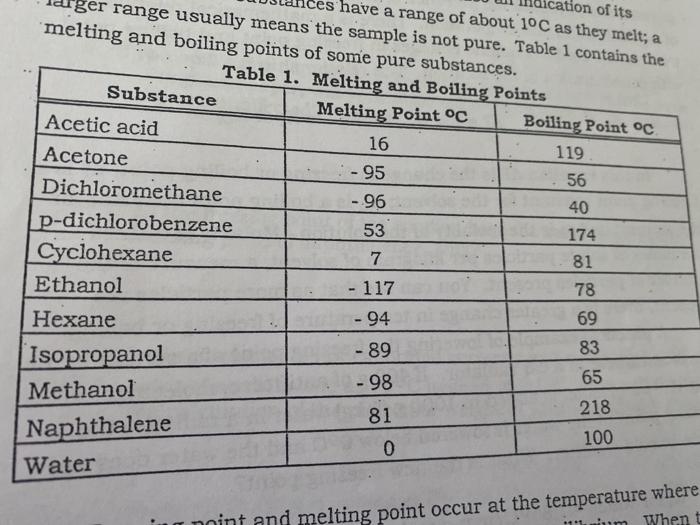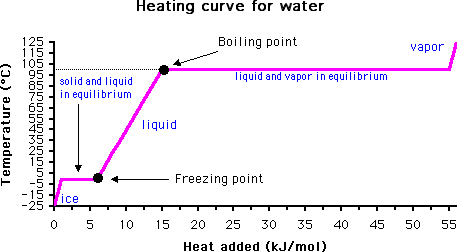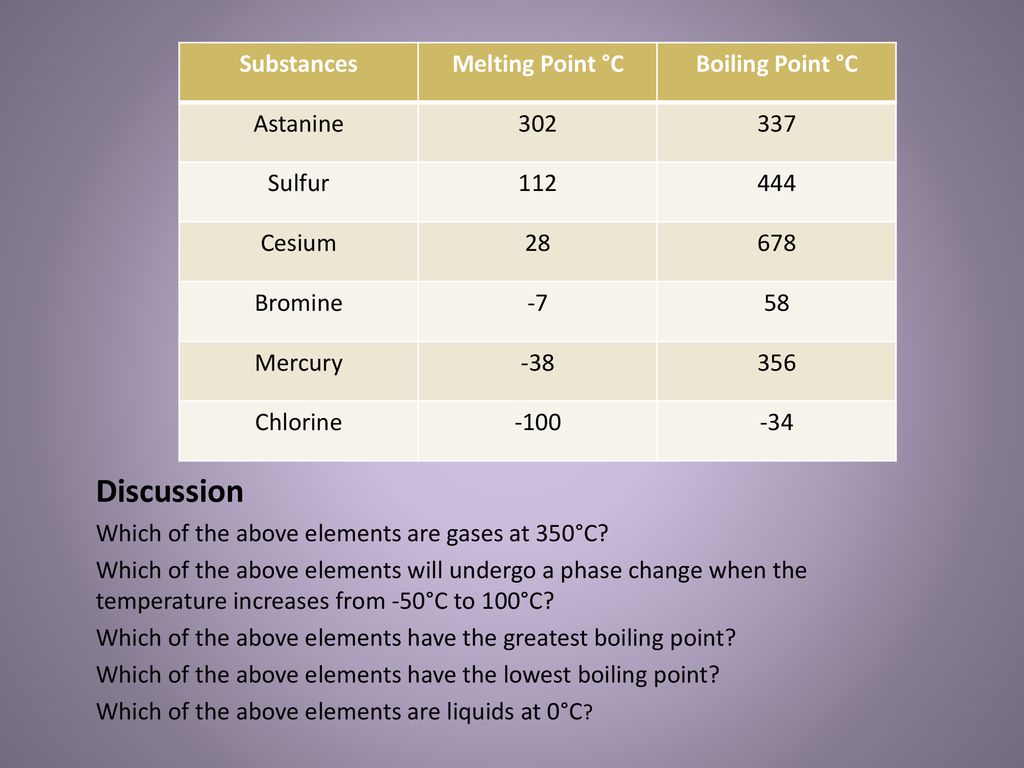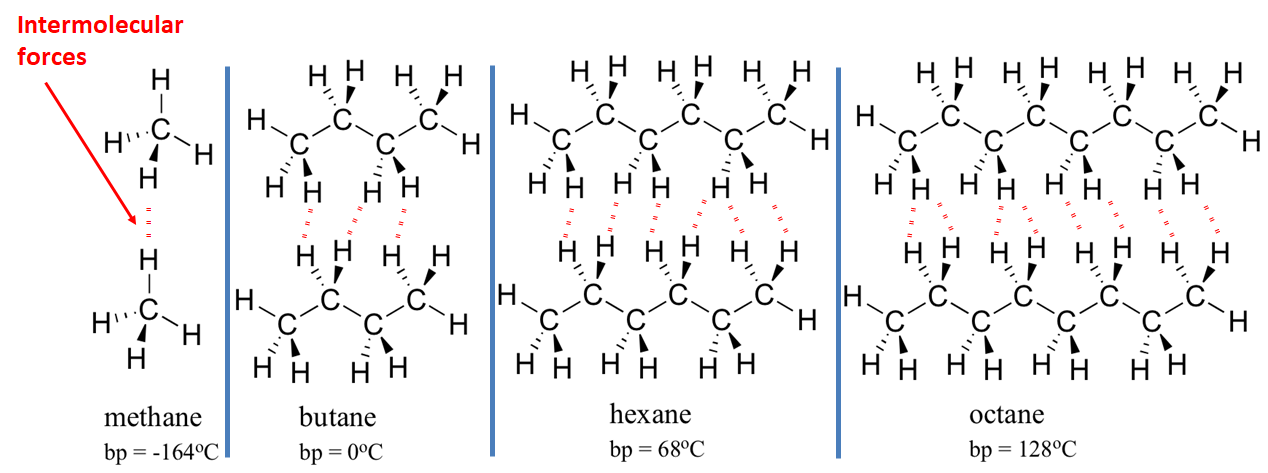
1:48 explain why the melting and boiling points of substances with simple molecular structures increase, in general, with increasing relative molecular mass - TutorMyself Chemistry
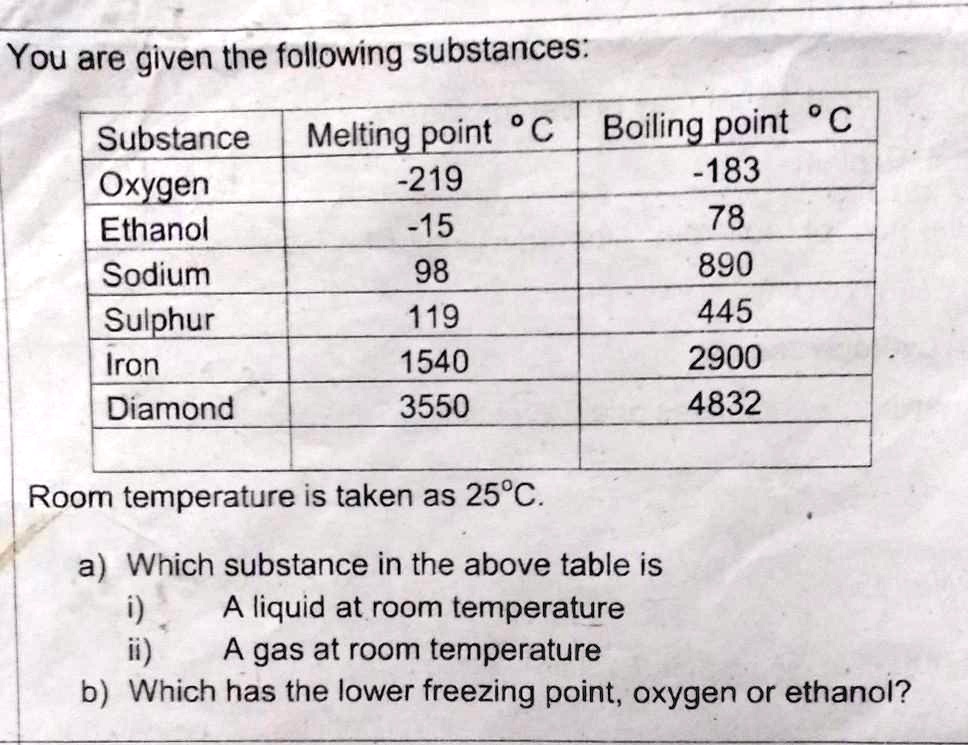
SOLVED: 'give the answer of of this question please You are given the following substances: point C Boiling point C Substance Melting -219 -183 Oxygen Ethanol -15 78 Sodium 98 890 Sulphur

Tackling Heat Energy Questions: Melting & Boiling Points! - Primary School Science Tuition - The Smart Student
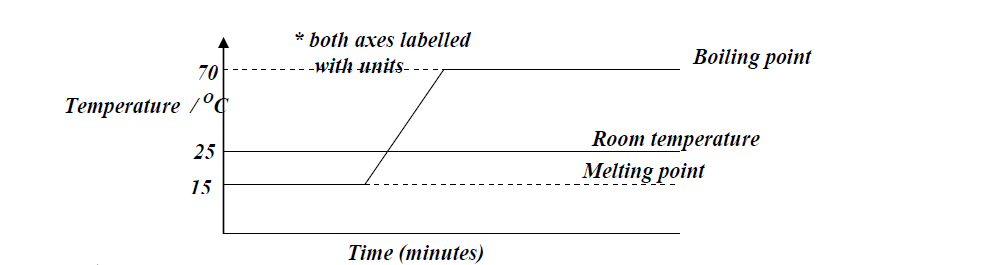
Substance Q has a melting point of 15°C and boiling point of 70°c. (a)On the same axis,draw the melting point and boiling point graph for Q...

SOLVED: ADVANCED MATERIAL Predicting the relative boiling points of pure substances point, 2 next to the substance with the next highest boiling point, and chemical symbol, chemical formula or Lewis structure substance

Tackling Heat Energy Questions: Melting & Boiling Points! - Primary School Science Tuition - The Smart Student
its normal boiling point is -189°C. Oxygen is a gas at room temperature. If the normal melting point of a substance is below room temperature, the substance is a liquid at room temperature. Benzene melts at 6°C and boils at 80°C; it is a liquid at room ...

Draw a phase diagram (pressure on the y axis, and temperature on the x-axis) from 0 atm to 2 atm and 0 ^oC to 200 ^oC for a substance that has a